| Pharmaceutical Information |
| Drug Name |
Deutetrabenazine |
| Drug ID |
BADD_D02507 |
| Description |
Deutetrabenazine is a novel, highly selective vesicular monoamine transporter 2 (VMAT2) inhibitor indicated for the management of chorea associated with Huntington’s disease. It is a hexahydro-dimethoxybenzoquinolizine derivative and a deuterated [DB04844] [A32046]. The presence of deuterium in deutetrabenazine increases the half-lives of the active metabolite and prolongs their pharmacological activity by attenuating CYP2D6 metabolism of the compound [A32046]. This allows less frequent dosing and a lower daily dose with improvement in tolerability [A32043]. Decreased plasma fluctuations of deutetrabenazine due to attenuated metabolism may explain a lower incidence of adverse reactions associated with deutetrabenazine [A32042]. Deutetrabenazine is a racemic mixture containing RR-Deutetrabenazine and SS-Deutetrabenazine [FDA Label].
Huntington's disease (HD) is a hereditary, progressive neurodegenerative disorder characterized by motor dysfunction, cognitive decline, and neuropsychiatric disturbances [A32043] that interfere with daily functioning and significantly reduce the quality of life. The most prominent physical symptom of HD that may increase the risk of injury is chorea, which is an involuntary, sudden movement that can affect any muscle and flow randomly across body regions [A32046]. Psychomotor symptoms of HD, such as chorea, are related to hyperactive dopaminergic neurotransmission [A14081]. Deutetrabenazine depletes the levels of presynaptic dopamine by blocking VMAT2, which is responsible for the uptake of dopamine into synaptic vesicles in monoaminergic neurons and exocytotic release [A14081]. As with other agents for the treatment of neurodegenerative diseases, deutetrabenazine is a drug to alleviate the motor symptoms of HD and is not proposed to halt the progression of the disease [T28]. In clinical trials of patients with HD, 12 weeks of treatment of deutetrabenazine resulted in overall improvement in mean total maximal chorea scores and motor signs than placebo [A32046]. It was approved by FDA in April 2017 and is marketed under the trade name Austedo as oral tablets. |
| Indications and Usage |
Indicated for the treatment of chorea associated with Huntington’s disease [FDA Label]. |
| Marketing Status |
approved; investigational |
| ATC Code |
N07XX16 |
| DrugBank ID |
DB12161
|
| KEGG ID |
D10701
|
| MeSH ID |
C000609690
|
| PubChem ID |
73437646
|
| TTD Drug ID |
D0E7ZU
|
| NDC Product Code |
59651-496; 68546-170; 68546-176; 53183-4062; 58032-2022; 68546-172; 68546-471; 68546-470; 68546-182; 68546-472; 68546-171; 68546-490; 65977-0123; 70600-025 |
| UNII |
P341G6W9NB
|
| Synonyms |
deutetrabenazine | Austedo |
|
| Chemical Information |
| Molecular Formula |
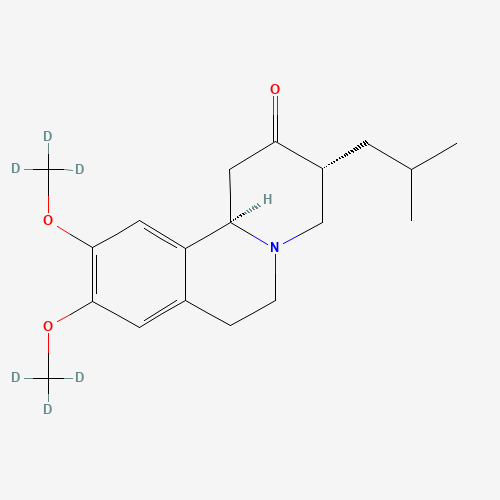
C19H27NO3 |
| CAS Registry Number |
1392826-25-3 |
| SMILES |
CC(C)CC1CN2CCC3=CC(=C(C=C3C2CC1=O)OC)OC |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice.
|
|
|

